A Comprehensive Study of 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine CAS 263401-62-3 in Heterocyclic Chemistry
3 min readIntroduction
In modern pharmaceutical and agrochemical research, heterocyclic compounds play a central role in the discovery and optimization of biologically active molecules. Among these, fused triazole–pyridazine derivatives have attracted significant attention due to their unique structural features and versatile chemical reactivity.
One such important compound is 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine, a specialized heterocyclic intermediate widely used in medicinal chemistry and advanced organic synthesis. Identified by CAS: 263401-62-3, this compound serves as a valuable building block in the development of new pharmaceutical candidates and functional materials.
This article provides a comprehensive overview of its chemical structure, physical properties, synthesis relevance, and industrial applications, along with its significance in modern drug discovery pipelines.
Chemical Identity and Structural Overview
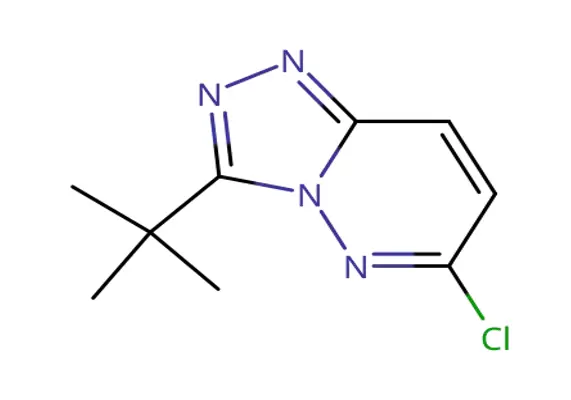
The compound 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine belongs to a class of fused bicyclic heterocycles composed of a triazole ring fused with a pyridazine ring. This structural arrangement provides both rigidity and electronic diversity, making it highly attractive in medicinal chemistry.
Key structural features include:
-
A [1,2,4]triazole ring, known for its pharmacological relevance
-
A pyridazine core, which contributes to electron-deficient aromaticity
-
A tert-butyl substituent at position 3, providing steric bulk and lipophilicity
-
A chlorine atom at position 6, enabling further chemical functionalization
The combination of electron-withdrawing and sterically demanding groups makes this molecule highly useful as a synthetic intermediate.
Molecular Properties
The physicochemical characteristics of this compound are essential for understanding its behavior in synthesis and formulation.
-
Chemical Name: 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine
-
CAS Number: 263401-62-3
-
Molecular Formula: C₉H₁₁ClN₄
-
Molecular Weight: 210.66 g/mol
-
Appearance: Typically a solid crystalline intermediate (depending on purity)
-
Purity: Commonly ≥98% in research-grade material
The presence of both heteroatoms (nitrogen and chlorine) and hydrophobic substituents results in a balanced polarity profile, making the compound suitable for a wide range of organic reactions.
Structural Significance in Medicinal Chemistry
Heterocyclic scaffolds such as triazolo-pyridazines are widely recognized in pharmaceutical research due to their ability to interact with biological targets, including enzymes, receptors, and nucleic acids.
The 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine scaffold is particularly interesting because:
1. Hydrogen Bonding Potential
The nitrogen atoms in the triazole and pyridazine rings can participate in hydrogen bonding interactions, which are essential for binding affinity in drug-target interactions.
2. Electron-Deficient Aromatic System
The fused heterocycle exhibits electron deficiency, allowing it to interact with nucleophilic sites in biological macromolecules.
3. Tunable Reactivity
The chlorine substituent at the 6-position serves as a reactive site for nucleophilic substitution, enabling structural diversification.
4. Lipophilicity Enhancement
The tert-butyl group increases hydrophobic character, improving membrane permeability in potential drug molecules.
These combined features make it a valuable scaffold for lead compound development.
Role in Organic Synthesis
In synthetic chemistry, this compound is primarily used as a heterocyclic building block. Its functional groups allow chemists to perform further derivatization and construct more complex molecules.
Common synthetic applications include:
-
Cross-coupling reactions (e.g., Suzuki, Buchwald–Hartwig)
-
Nucleophilic substitution at the chloro position
-
Scaffold diversification for structure–activity relationship (SAR) studies
-
Intermediate formation in multi-step drug synthesis routes
Because of its reactive chlorine atom and stable fused ring system, it is often used as a key intermediate rather than a final product.
Pharmaceutical Research Applications
Although not a drug itself, 3-tert-butyl-6-chloro-[1,2,4]triazolo[4,3-b]pyridazine is frequently used in early-stage drug discovery programs.
Potential therapeutic areas influenced by similar scaffolds include:
1. Anticancer Research
Triazole-containing compounds often show activity against kinase targets involved in tumor growth.
2. Antimicrobial Agents
Heterocyclic systems can disrupt bacterial enzyme function or cell wall synthesis pathways.
3. CNS-Active Compounds
Pyridazine derivatives have been explored for neurological receptor modulation.
4. Anti-inflammatory Agents
Structural analogs are studied for inhibition of inflammatory mediators.
About Author

